Transcranial magnetic stimulation
| Transcranial magnetic stimulation | |
|---|---|
 Transcranial magnetic stimulation (schematic diagram) | |
| Specialty | Psychiatry, neurology |
| MeSH | D050781 |
Transcranial magnetic stimulation (TMS) is a noninvasive form of brain stimulation in which a changing magnetic field is used to induce an electric current at a specific area of the brain through electromagnetic induction. An electric pulse generator, or stimulator, is connected to a magnetic coil connected to the scalp. The stimulator generates a changing electric current within the coil which creates a varying magnetic field, inducing a current within a region in the brain itself.[1]: 3 [2]
TMS has shown diagnostic and therapeutic potential in the central nervous system with a wide variety of disease states in neurology and mental health, but has no demonstrated clinical worth for treatment of any other condition.[3]
Adverse effects of TMS appear rare and include fainting and seizure.[4] Other potential issues include discomfort, pain, hypomania, cognitive change, hearing loss, and inadvertent current induction in implanted devices such as pacemakers or defibrillators.[4]
Medical uses
[edit]
TMS does not require surgery or electrode implantation.
Its use can be diagnostic and/or therapeutic. Effects vary based on frequency and intensity of the magnetic pulses as well as the length of treatment, which dictates the total number of pulses given.[6] TMS treatments are approved by the FDA in the US and by NICE in the UK for the treatment of depression and are provided by private clinics and some VA medical centers. TMS stimulates cortical tissue without the pain sensations produced in transcranial electrical stimulation.[7]
Diagnosis
[edit]TMS can be used clinically to measure activity and function of specific brain circuits in humans, most commonly with single or paired magnetic pulses.[8] The most widely accepted use is in measuring the connection between the primary motor cortex of the central nervous system and the peripheral nervous system to evaluate damage related to past or progressive neurologic insult.[8][9][10][11] TMS has utility as a diagnostic instrument for myelopathy, amyotrophic lateral sclerosis, and multiple sclerosis.[12]
Treatment
[edit]There is some evidence that TMS may have applications for a number of conditions including depression, fibromyalgia and neuropathic pain, and TMS treatment is covered by most private insurance plans as well as by traditional Medicare, but for no condition does the evidence rise to the level of showing clinical relevance.[3]
Adverse effects
[edit]Although TMS is generally regarded as safe, risks are increased for therapeutic rTMS compared to single or paired diagnostic TMS.[13] Adverse effects generally increase with higher frequency stimulation.[4]
The greatest immediate risk from TMS is fainting, though this is uncommon. Seizures have been reported, but are rare.[4][14][15] Other adverse effects include short term discomfort, pain, brief episodes of hypomania, cognitive change, hearing loss, impaired working memory, and the induction of electrical currents in implanted devices such as cardiac pacemakers.[4]
Procedure
[edit]During the procedure, a magnetic coil is positioned at the head of the person receiving the treatment using anatomical landmarks on the skull, in particular the inion and nasion.[5] The coil is then connected to a pulse generator, or stimulator, that delivers electric current to the coil.[2]
Physics
[edit]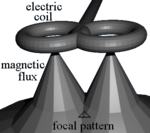

TMS uses electromagnetic induction to generate an electric current across the scalp and skull.[16][17] A plastic-enclosed coil of wire is held next to the skull and when activated, produces a varying magnetic field oriented orthogonally to the plane of the coil. The changing magnetic field then induces an electric current in the brain that activates nearby nerve cells in a manner similar to a current applied superficially at the cortical surface.[18]
The magnetic field is about the same strength as magnetic resonance imaging (MRI), and the pulse generally reaches no more than 5 centimeters into the brain unless using a modified coil and technique for deeper stimulation.[17]
Transcranial magnetic stimulation is achieved by quickly discharging current from a large capacitor into a coil to produce pulsed magnetic fields between 2 and 3 teslas in strength.[19] Directing the magnetic field pulse at a targeted area in the brain causes a localized electrical current which can then either depolarize or hyperpolarize neurons at that site. The induced electric field inside the brain tissue causes a change in transmembrane potentials resulting in depolarization or hyperpolarization of neurons, causing them to be more or less excitable, respectively.[19]
TMS usually stimulates to a depth from 2 to 4 cm below the surface, depending on the coil and intensity used. Consequently, only superficial brain areas can be affected.[20] Deep TMS can reach up to 6 cm into the brain to stimulate deeper layers of the motor cortex, such as that which controls leg motion. The path of this current can be difficult to model because the brain is irregularly shaped with variable internal density and water content, leading to a nonuniform magnetic field strength and conduction throughout its tissues.[21]
Frequency and duration
[edit]The effects of TMS can be divided based on frequency, duration and intensity (amplitude) of stimulation:[22]
- Single or paired pulse TMS causes neurons in the neocortex under the site of stimulation to depolarize and discharge an action potential. If used in the primary motor cortex, it produces muscle activity referred to as a motor evoked potential (MEP) which can be recorded on electromyography. If used on the occipital cortex, 'phosphenes' (flashes of light) might be perceived by the subject. In most other areas of the cortex, there is no conscious effect, but behaviour may be altered (e.g., slower reaction time on a cognitive task), or changes in brain activity may be detected using diagnostic equipment.[23]
- Repetitive TMS produces longer-lasting effects which persist past the period of stimulation. rTMS can increase or decrease the excitability of the corticospinal tract depending on the intensity of stimulation, coil orientation, and frequency. Low frequency rTMS with a stimulus frequency less than 1 Hz is believed to inhibit cortical firing while a stimulus frequency greater than 1 Hz, or high frequency, is believed to provoke it.[24] Though its mechanism is not clear, it has been suggested as being due to a change in synaptic efficacy related to long-term potentiation (LTP) and long-term depression like plasticity (LTD-like plasticity).[25][26]
Coil types
[edit]Most devices use a coil shaped like a figure-eight to deliver a shallow magnetic field that affects more superficial neurons in the brain.[27] Differences in magnetic coil design are considered when comparing results, with important elements including the type of material, geometry and specific characteristics of the associated magnetic pulse.
The core material may be either a magnetically inert substrate ('air core'), or a solid, ferromagnetically active material ('solid core'). Solid cores result in more efficient transfer of electrical energy to a magnetic field and reduce energy loss to heat, and so can be operated with the higher volume of therapy protocols without interruption due to overheating. Varying the geometric shape of the coil itself can cause variations in focality, shape, and depth of penetration. Differences in coil material and its power supply also affect magnetic pulse width and duration.[28]
A number of different types of coils exist, each of which produce different magnetic fields. The round coil is the original used in TMS. Later, the figure-eight (butterfly) coil was developed to provide a more focal pattern of activation in the brain, and the four-leaf coil for focal stimulation of peripheral nerves. The double-cone coil conforms more to the shape of the head.[29] The Hesed (H-core), circular crown and double cone coils allow more widespread activation and a deeper magnetic penetration. They are supposed to impact deeper areas in the motor cortex and cerebellum controlling the legs and pelvic floor, for example, though the increased depth comes at the cost of a less focused magnetic pulse.[4]
Research directions
[edit]For Parkinson's disease, early results suggest that low frequency stimulation may have an effect on medication associated dyskinesia, and that high frequency stimulation improves motor function.[30][31]
History
[edit]Luigi Galvani (1737–1798) undertook research on the effects of electricity on the body in the late-eighteenth century and laid the foundations for the field of electrophysiology.[32] In the 1830s Michael Faraday (1791–1867) discovered that an electrical current had a corresponding magnetic field, and that changing one could induce its counterpart.[33]
Work to directly stimulate the human brain with electricity started in the late 1800s, and by the 1930s the Italian physicians Cerletti and Bini had developed electroconvulsive therapy (ECT).[32] ECT became widely used to treat mental illness, and ultimately overused, as it began to be seen as a panacea. This led to a backlash in the 1970s.[32]
In 1980 Merton and Morton successfully used transcranial electrical stimulation (TES) to stimulate the motor cortex. However, this process was very uncomfortable, and subsequently Anthony T. Barker began to search for an alternative to TES.[34] He began exploring the use of magnetic fields to alter electrical signaling within the brain, and the first stable TMS devices were developed in 1985.[32][33] They were originally intended as diagnostic and research devices, with evaluation of their therapeutic potential being a later development.[32][33] The United States' FDA first approved TMS devices in October 2008.[32]
Regulatory status
[edit]Speech mapping prior to neurosurgery
[edit]Nexstim obtained United States Federal Food, Drug, and Cosmetic Act§Section 510(k) clearance for the assessment of the primary motor cortex for pre-procedural planning in December 2009[35] and for neurosurgical planning in June 2011.[36]
Depression
[edit]TMS is approved as a Class II medical device under the "de novo pathway".[37][38]
Obsessive–compulsive disorder (OCD)
[edit]In August 2018, the US Food and Drug Administration (US FDA) authorized the use of TMS developed by the Israeli company Brainsway in the treatment of obsessive–compulsive disorder (OCD).[39]
In 2020, US FDA authorized the use of TMS developed by the U.S. company MagVenture Inc. in the treatment of OCD.[40]
In 2023, US FDA authorized the use of TMS developed by the U.S. company Neuronetics Inc. in the treatment of OCD.[41]
Other neurological areas
[edit]In the European Economic Area, various versions of deep TMS H-coils have CE marking for Alzheimer's disease,[42] autism,[42] bipolar disorder,[43] epilepsy,[44] chronic pain,[43] major depressive disorder,[43] Parkinson's disease,[45][46] post-traumatic stress disorder (PTSD),[43][47] schizophrenia (negative symptoms)[43] and to aid smoking cessation.[42] One review found tentative benefit for cognitive enhancement in healthy people.[48]
Coverage by health services and insurers
[edit]United Kingdom
[edit]The United Kingdom's National Institute for Health and Care Excellence (NICE) issues guidance to the National Health Service (NHS) in England, Wales, Scotland and Northern Ireland (UK). NICE guidance does not cover whether or not the NHS should fund a procedure. Local NHS bodies (primary care trusts and hospital trusts) make decisions about funding after considering the clinical effectiveness of the procedure and whether the procedure represents value for money for the NHS.[49]
NICE evaluated TMS for severe depression (IPG 242) in 2007, and subsequently considered TMS for reassessment in January 2011 but did not change its evaluation.[50] The Institute found that TMS is safe, but there is insufficient evidence for its efficacy.[50]
In January 2014, NICE reported the results of an evaluation of TMS for treating and preventing migraine (IPG 477). NICE found that short-term TMS is safe but there is insufficient evidence to evaluate safety for long-term and frequent uses. It found that evidence on the efficacy of TMS for the treatment of migraine is limited in quantity, that evidence for the prevention of migraine is limited in both quality and quantity.[51]
Subsequently, in 2015, NICE approved the use of TMS for the treatment of depression in the UK and IPG542 replaced IPG242.[52] NICE said "The evidence on repetitive transcranial magnetic stimulation for depression shows no major safety concerns. The evidence on its efficacy in the short-term is adequate, although the clinical response is variable. Repetitive transcranial magnetic stimulation for depression may be used with normal arrangements for clinical governance and audit."
United States: commercial health insurance
[edit]In 2013, several commercial health insurance plans in the United States, including Anthem, Health Net, Kaiser Permanente, and Blue Cross Blue Shield of Nebraska and of Rhode Island, covered TMS for the treatment of depression for the first time.[53][54][55][56] In contrast, UnitedHealthcare issued a medical policy for TMS in 2013 that stated there is insufficient evidence that the procedure is beneficial for health outcomes in patients with depression. UnitedHealthcare noted that methodological concerns raised about the scientific evidence studying TMS for depression include small sample size, lack of a validated sham comparison in randomized controlled studies, and variable uses of outcome measures.[57] Other commercial insurance plans whose 2013 medical coverage policies stated that the role of TMS in the treatment of depression and other disorders had not been clearly established or remained investigational included Aetna, Cigna and Regence.[58][59][60]
United States: Medicare
[edit]Policies for Medicare coverage vary among local jurisdictions within the Medicare system,[61] and Medicare coverage for TMS has varied among jurisdictions and with time. For example:
- In early 2012 in New England, Medicare covered TMS for the first time in the United States.[62][63][64][65] However, that jurisdiction later decided to end coverage after October, 2013.[66]
- In August 2012, the jurisdiction covering Arkansas, Louisiana, Mississippi, Colorado, Texas, Oklahoma, and New Mexico determined that there was insufficient evidence to cover the treatment,[67] but the same jurisdiction subsequently determined that Medicare would cover TMS for the treatment of depression after December 2013.[68]
Limitations
[edit]There are serious concerns about stimulating brain tissue using non-invasive magnetic field methods:
First, the uncertainty in the dose (time and technical field parameters) for correct and healthy stimulation is a complex and challenging issue.[69] While neurophysiology lacks knowledge about the nature of such a treatment of nervous diseases at the cellular level,[70] this method involves excessive exposure of the brain to an intense field, which is several times and even orders of magnitude higher than natural electromagnetic fields in the brain.[71][72]
Second, it is impossible to localize the effect of stimulation on specific neural networks that need to be treated.[73] Neuronal activity related to mental processes, neuronal correlates of cognitive functions are still intriguing questions for contemporary research. Non-invasive brain tissue stimulation targets a large area of poorly characterized tissue. An undefined dose and target of radiation can destroy healthy cells during a treatment procedure. Because it is unclear whether magnetic fields reach only those neural structures of the brain that need treatment, this uncertainty challenges the selection of treatment for neurological disorders by magnetic fields. Thus, magnetic field exposure may destroy healthy neuronal structures. The inability to localize the effect of stimulation makes it challenging to target stimulation only to the desired neural networks. [73]
See also
[edit]- Cortical stimulation mapping
- Cranial electrotherapy stimulation
- Electrical brain stimulation
- Electroconvulsive therapy
- Low field magnetic stimulation
- My Beautiful Broken Brain
- Non-invasive cerebellar stimulation
- Transcranial alternating current stimulation
- Transcranial direct-current stimulation
- Transcranial random noise stimulation
- Vagus nerve stimulation
References
[edit]- ^ NICE. January 2014 Transcranial magnetic stimulation for treating and preventing migraine
- ^ a b Michael Craig Miller for Harvard Health Publications. July 26, 2012 Magnetic stimulation: a new approach to treating depression?
- ^ a b Lefaucheur JP, Aleman A, Baeken C, Benninger DH, Brunelin J, et al. (February 2020). "Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS): An update (2014-2018)". Clin Neurophysiol. 131 (2): 474–528. doi:10.1016/j.clinph.2019.11.002. hdl:10362/147799. PMID 31901449.
The present work only provides arguments to be confident that some rTMS protocols do something that is different from a placebo in some indications, but not that the results obtained are clinically relevant
- ^ a b c d e f Rossi; et al. (January 2021). "Safety and recommendations for TMS use in healthy subjects and patient populations, with updates on training, ethical and regulatory issues: Expert Guidelines". Clinical Neurophysiology. 132 (1): 269–306. doi:10.1016/j.clinph.2020.10.003. PMC 9094636. PMID 33243615. S2CID 225049093.
- ^ a b Nauczyciel C, Hellier P, Morandi X, Blestel S, Drapier D, Ferre JC, et al. (April 2011). "Assessment of standard coil positioning in transcranial magnetic stimulation in depression". Psychiatry Research. 186 (2–3): 232–8. doi:10.1016/j.psychres.2010.06.012. PMID 20692709. S2CID 25100990.
- ^ Klomjai, Wanalee; Katz, Rose; Lackmy-Vallée, Alexandra (2015-09-01). "Basic principles of transcranial magnetic stimulation (TMS) and repetitive TMS (rTMS)". Annals of Physical and Rehabilitation Medicine. 58 (4): 208–213. doi:10.1016/j.rehab.2015.05.005. ISSN 1877-0657. PMID 26319963.
- ^ Lefaucher, Jean-Pascal (2019). "37. Transcranial magnetic stimulation". In Levin, Kerry H.; Chauvel, Patrick (eds.). Clinical Neurophysiology: Basis and Technical Aspects. Handbook of Clinical Neurology. Vol. 160. Elsevier. pp. 559–580. doi:10.1016/B978-0-444-64032-1.00037-0. ISBN 978-0-444-64032-1.
- ^ a b Groppa S, Oliviero A, Eisen A, Quartarone A, Cohen LG, Mall V, et al. (May 2012). "A practical guide to diagnostic transcranial magnetic stimulation: report of an IFCN committee". Clinical Neurophysiology. 123 (5): 858–882. doi:10.1016/j.clinph.2012.01.010. PMC 4890546. PMID 22349304.
- ^ Rossini PM, Rossi S (February 2007). "Transcranial magnetic stimulation: diagnostic, therapeutic, and research potential". Neurology. 68 (7): 484–8. doi:10.1212/01.wnl.0000250268.13789.b2. PMID 17296913. S2CID 19629888.
- ^ Dimyan MA, Cohen LG (February 2010). "Contribution of transcranial magnetic stimulation to the understanding of functional recovery mechanisms after stroke". Neurorehabilitation and Neural Repair. 24 (2): 125–135. doi:10.1177/1545968309345270. PMC 2945387. PMID 19767591.
- ^ Nowak DA, Bösl K, Podubeckà J, Carey JR (2010). "Noninvasive brain stimulation and motor recovery after stroke". Restorative Neurology and Neuroscience. 28 (4): 531–544. doi:10.3233/RNN-2010-0552. PMID 20714076.
- ^ Chen R, Cros D, Curra A, Di Lazzaro V, Lefaucheur JP, Magistris MR, Mills K, Rösler KM, Triggs WJ, Ugawa Y, Ziemann U (March 2008). "The clinical diagnostic utility of transcranial magnetic stimulation: report of an IFCN committee". Clin Neurophysiol. 119 (3): 504–532. doi:10.1016/j.clinph.2007.10.014. PMID 18063409.
- ^ van den Noort M, Lim S, Bosch P (December 2014). "Recognizing the risks of brain stimulation". Science. 346 (6215): 1307. doi:10.1126/science.346.6215.1307-a. hdl:2066/133976. PMID 25504707.
- ^ Dobek CE, Blumberger DM, Downar J, Daskalakis ZJ, Vila-Rodriguez F (2015). "Risk of seizures in transcranial magnetic stimulation: a clinical review to inform consent process focused on bupropion". Neuropsychiatric Disease and Treatment. 11: 2975–87. doi:10.2147/NDT.S91126. PMC 4670017. PMID 26664122.
- ^ Fitzgerald PB, Daskalakis ZJ (2013). "7. rTMS-Associated Adverse Events". Repetitive Transcranial Magnetic Stimulation for Depressive Disorders. Springer. pp. 81–90. doi:10.1007/978-3-642-36467-9_7. ISBN 978-3-642-36466-2.
- ^ Cavaleri R, Schabrun SM, Chipchase LS (March 2017). "The number of stimuli required to reliably assess corticomotor excitability and primary motor cortical representations using transcranial magnetic stimulation (TMS): a systematic review and meta-analysis". Systematic Reviews. 6 (1): 48. doi:10.1186/s13643-017-0440-8. PMC 5340029. PMID 28264713.
- ^ a b "Brain Stimulation Therapies". NIMH.
- ^ Cacioppo JT, Tassinary LG, Berntson GG, eds. (2007). Handbook of psychophysiology (3rd ed.). Cambridge University Press. p. 121. doi:10.1017/CBO9780511546396. ISBN 978-0-521-84471-0.
- ^ a b Walsh, V.; Pascual-Leone, A. (2003). Transcranial Magnetic Stimulation: A Neurochronometrics of Mind. Cambridge, MA: MIT Press. doi:10.7551/mitpress/6896.001.0001. ISBN 978-0-262-28573-5. OCLC 53889320.
- ^ Lefaucheur JP, André-Obadia N, Antal A, Ayache SS, Baeken C, Benninger DH, Cantello RM, Cincotta M, de Carvalho M, De Ridder D, Devanne H, Di Lazzaro V, Filipović SR, Hummel FC, Jääskeläinen SK, Kimiskidis VK, Koch G, Langguth B, Nyffeler T, Oliviero A, Padberg F, Poulet E, Rossi S, Rossini PM, Rothwell JC, Schönfeldt-Lecuona C, Siebner HR, Slotema CW, Stagg CJ, Valls-Sole J, Ziemann U, Paulus W, Garcia-Larrea L (November 2014). "Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS)". Clin Neurophysiol. 125 (11): 2150–2206. doi:10.1016/j.clinph.2014.05.021. PMID 25034472.
- ^ See:
- Zangen A, Roth Y, Voller B, Hallett M (April 2005). "Transcranial magnetic stimulation of deep brain regions: evidence for efficacy of the H-coil". Clinical Neurophysiology. 116 (4): 775–9. doi:10.1016/j.clinph.2004.11.008. PMID 15792886. S2CID 25101101.
- Huang YZ, Sommer M, Thickbroom G, Hamada M, Pascual-Leonne A, Paulus W, et al. (January 2009). "Consensus: New methodologies for brain stimulation". Brain Stimulation. 2 (1): 2–13. doi:10.1016/j.brs.2008.09.007. PMC 5507351. PMID 20633398.
- ^ Rubens MT, Zanto TP (March 2012). "Parameterization of transcranial magnetic stimulation". Journal of Neurophysiology. 107 (5): 1257–9. doi:10.1152/jn.00716.2011. PMC 3311692. PMID 22072509.
- ^ Pascual-Leone A, Davey N, Rothwell J, Wassermann EM, Puri BK (2002). Handbook of Transcranial Magnetic Stimulation. London: Edward Arnold. ISBN 978-0-340-72009-7. OCLC 47824021.
- ^ Cusin C, Dougherty DD (August 2012). "Somatic therapies for treatment-resistant depression: ECT, TMS, VNS, DBS". Biology of Mood & Anxiety Disorders. 2 (1): 14. doi:10.1186/2045-5380-2-14. PMC 3514332. PMID 22901565.
- ^ Fitzgerald PB, Fountain S, Daskalakis ZJ (December 2006). "A comprehensive review of the effects of rTMS on motor cortical excitability and inhibition". Clinical Neurophysiology. 117 (12): 2584–96. doi:10.1016/j.clinph.2006.06.712. PMID 16890483. S2CID 31458874.
- ^ Baur D, Galevska D, Hussain S, Cohen LG, Ziemann U, Zrenner C (2020). "Induction of LTD-like corticospinal plasticity by low-frequency rTMS depends on pre-stimulus phase of sensorimotor μ-rhythm". Brain Stimul. 13 (6): 1580–7. doi:10.1016/j.brs.2020.09.005. PMC 7710977. PMID 32949780.
- ^ Bersani, F.S.; Minichino, A.; Enticott, P.G.; Mazzarini, L.; Khan, N.; Antonacci, G.; Raccah, R.N.; Salviati, M.; Delle Chiaie, R.; Bersani, G.; Fitzgerald, P.B.; Biondi, M. (January 2013). "Deep transcranial magnetic stimulation as a treatment for psychiatric disorders: A comprehensive review". European Psychiatry. 28 (1): 30–39. doi:10.1016/j.eurpsy.2012.02.006. PMID 22559998. S2CID 29053871.
- ^ Riehl M (2008). "TMS Stimulator Design". In Wassermann EM, Epstein CM, Ziemann U, Walsh V, Paus T, Lisanby SH (eds.). Oxford Handbook of Transcranial Stimulation. Oxford: Oxford University Press. pp. 13–23, 25–32. doi:10.1093/oxfordhb/9780198568926.013.0003. ISBN 978-0-19-856892-6.
- ^ Roth BJ, Maccabee PJ, Eberle LP, Amassian VE, Hallett M, Cadwell J, et al. (February 1994). "In vitro evaluation of a 4-leaf coil design for magnetic stimulation of peripheral nerve". Electroencephalography and Clinical Neurophysiology. 93 (1): 68–74. doi:10.1016/0168-5597(94)90093-0. PMID 7511524.
- ^ Machado S, Bittencourt J, Minc D, Portella CE, Velasques B, Cunha M, et al. (2008). "Therapeutic applications of repetitive transcranial magnetic stimulation in clinical neurorehabilitation". Functional Neurology. 23 (3): 113–122. PMID 19152730.
- ^ Liu, Xuan; Li, Lei; Liu, Ye (2023-09-29). "Comparative motor effectiveness of non-invasive brain stimulation techniques in patients with Parkinson's disease: A network meta-analysis". Medicine. 102 (39): e34960. doi:10.1097/MD.0000000000034960. ISSN 1536-5964. PMC 10545289. PMID 37773851.
- ^ a b c d e f Horvath JC, Perez JM, Forrow L, Fregni F, Pascual-Leone A (March 2011). "Transcranial magnetic stimulation: a historical evaluation and future prognosis of therapeutically relevant ethical concerns". Journal of Medical Ethics. 37 (3): 137–143. doi:10.1136/jme.2010.039966. JSTOR 23034661. PMID 21106996. S2CID 13262044.
- ^ a b c Noohi S, Amirsalari S (2016). "History, Studies and Specific Uses of Repetitive Transcranial Magnetic Stimulation (rTMS) in Treating Epilepsy". Iranian Journal of Child Neurology. 10 (1): 1–8. PMC 4815479. PMID 27057180.
- ^ Klomjai W, Katz R, Lackmy-Vallée A (September 2015). "Basic principles of transcranial magnetic stimulation (TMS) and repetitive TMS (rTMS)". Annals of Physical and Rehabilitation Medicine. 58 (4): 208–213. doi:10.1016/j.rehab.2015.05.005. PMID 26319963.
- ^ "FDA clears Nexstim´s Navigated Brain Stimulation for non-invasive cortical mapping prior to neurosurgery – Archive – Press Releases". nexstim.com.
- ^ "Nexstim Announces FDA Clearance for NexSpeech® – Enabling Noninvasive Speech Mapping Prior to Neurosurgery". businesswire.com. 11 June 2012.
- ^ Michael Drues, for Med Device Online. 5 February 2014 Secrets Of The De Novo Pathway, Part 1: Why Aren't More Device Makers Using It?
- ^ Schwedt TJ, Vargas B (September 2015). "Neurostimulation for Treatment of Migraine and Cluster Headache". Pain Medicine. 16 (9): 1827–34. doi:10.1111/pme.12792. PMC 4572909. PMID 26177612.
- ^ "FDA permits marketing of transcranial magnetic stimulation for treatment of obsessive compulsive disorder". Food and Drug Administration. 2020-02-20.
- ^ "MagVenture receives FDA clearance for OCD | Clinical TMS Society". www.clinicaltmssociety.org. Retrieved 2023-10-11.
- ^ "FDA clears OCD motor threshold cap for transcranial magnetic stimulation system". www.healio.com. Retrieved 2023-10-11.
- ^ a b c "Brainsway reports positive Deep TMS system trial data for OCD". Medical Device Network. Medicaldevice-network. September 6, 2013. Retrieved December 16, 2013.
- ^ a b c d e "Brainsway's Deep TMS EU Cleared for Neuropathic Chronic Pain". medGadget. July 3, 2012. Retrieved December 16, 2013.
- ^ Gersner R, Oberman L, Sanchez MJ, Chiriboga N, Kaye HL, Pascual-Leone A, et al. (2016-01-01). "H-coil repetitive transcranial magnetic stimulation for treatment of temporal lobe epilepsy: A case report". Epilepsy & Behavior Case Reports. 5 (Supplement C): 52–56. doi:10.1016/j.ebcr.2016.03.001. PMC 4832041. PMID 27114902.
- ^ Chou YH, Hickey PT, Sundman M, Song AW, Chen NK (April 2015). "Effects of repetitive transcranial magnetic stimulation on motor symptoms in Parkinson disease: a systematic review and meta-analysis". JAMA Neurology. 72 (4): 432–440. doi:10.1001/jamaneurol.2014.4380. PMC 4425190. PMID 25686212.
- ^ Torres F, Villalon E, Poblete P, Moraga-Amaro R, Linsambarth S, Riquelme R, et al. (2015-10-26). "Retrospective Evaluation of Deep Transcranial Magnetic Stimulation as Add-On Treatment for Parkinson's Disease". Frontiers in Neurology. 6: 210. doi:10.3389/fneur.2015.00210. PMC 4620693. PMID 26579065.
- ^ Petrosino, Nicholas J.; Cosmo, Camila; Berlow, Yosef A.; Zandvakili, Amin; van ’t Wout-Frank, Mascha; Philip, Noah S. (2021). "Transcranial magnetic stimulation for post-traumatic stress disorder". Therapeutic Advances in Psychopharmacology. 11. doi:10.1177/20451253211049921. PMC 8558793. PMID 34733479.
- ^ Luber B, Lisanby SH (January 2014). "Enhancement of human cognitive performance using transcranial magnetic stimulation (TMS)". NeuroImage. 85 Pt 3 (3): 961–970. doi:10.1016/j.neuroimage.2013.06.007. PMC 4083569. PMID 23770409.
- ^ NICE About NICE: What we do
- ^ a b "Transcranial magnetic stimulation for severe depression (IPG242)". London: National Institute for Health and Clinical Excellence. 2011-03-04.
- ^ "Transcranial magnetic stimulation for treating and preventing migraine". London: National Institute for Health and Clinical Excellence. January 2014.
- ^ "Repetitive transcranial magnetic stimulation for depression". National Institute for Health and Care Excellence. 16 December 2015. Retrieved 6 December 2019.
- ^ "Medical Policy: Transcranial Magnetic Stimulation for Depression and Other Neuropsychiatric Disorders". Policy No. BEH.00002. Anthem, Inc. 2013-04-16. Archived from the original on 2013-07-29. Retrieved 2013-12-11.
- ^ Health Net (March 2012). "National Medical Policy: Transcranial Magnetic Stimulation" (PDF). Policy Number NMP 508. Health Net. Archived from the original (PDF) on 2012-10-11. Retrieved 2012-09-05.
- ^ "Medical Policy Manual" (PDF). Section IV.67. Blue Cross Blue Shield of Nebraska. 2011-05-18. Archived from the original (PDF) on 2012-10-28.
- ^ "Medical Coverage Policy: Transcranial Magnetic Stimulation for Treatment of Depression and Other Psychiatric/Neurologic Disorders" (PDF). Blue Cross Blue Shield of Rhode Island. 2012-05-15. Archived from the original (PDF) on 2013-05-26. Retrieved 2012-09-05.
- ^ UnitedHealthcare (2013-12-01). "Transcranial Magnetic Stimulation" (PDF). UnitedHealthCare. p. 2. Archived from the original (PDF) on 2013-05-20. Retrieved 2013-12-11.
- ^ Aetna (2013-10-11). "Clinical Policy Bulletin: Transcranial Magnetic Stimulation and Cranial Electrical Stimulation". Number 0469. Aetna. Archived from the original on 2013-10-22. Retrieved 2013-12-11.
- ^ Cigna (2013-01-15). "Cigna Medical Coverage Policy: Transcranial Magnetic Stimulation" (PDF). Coverage Policy Number 0383. Cigna. Archived from the original (PDF) on 2017-02-04. Retrieved 2013-12-11.
- ^ Regence (2013-06-01). "Medical Policy: Transcranial Magnetic Stimulation as a Treatment of Depression and Other Disorders" (PDF). Policy No. 17. Regence. Archived from the original (PDF) on 2014-12-09. Retrieved 2013-12-11.
- ^ "Medicare Administrative Contractors". Centers for Medicare and Medicaid Services. 2013-07-10. Archived from the original on 2014-02-14. Retrieved 2014-02-14.
- ^ NHIC, Corp. (2013-10-24). "Local Coverage Determination (LCD) for Repetitive Transcranial Magnetic Stimulation (rTMS) (L32228)". Centers for Medicare and Medicaid Services. Archived from the original on 2014-02-17. Retrieved 2014-02-17.
- ^ "Important Treatment Option for Depression Receives Medicare Coverage". Press Release. PBN.com: Providence Business News. 2012-03-30. Archived from the original on 2013-04-05. Retrieved 2012-10-11.
- ^ The Institute for Clinical and Economic Review (June 2012). "Coverage Policy Analysis: Repetitive Transcranial Magnetic Stimulation (rTMS)" (PDF). The New England Comparative Effectiveness Public Advisory Council (CEPAC). Archived from the original (PDF) on 2013-12-13. Retrieved 2013-12-11.
- ^ "Transcranial Magnetic Stimulation Cites Influence of New England Comparative Effectiveness Public Advisory Council (CEPAC)". Berlin, Vermont: Central Vermont Medical Center. 2012-02-06. Archived from the original on 2012-03-25. Retrieved 2012-10-12.
- ^ National Government Services, Inc. (2013-10-25). "Local Coverage Determination (LCD): Transcranial Magnetic Stimulation (L32038)". Centers for Medicare and Medicaid Services. Retrieved 2014-02-17.
- ^ Novitas Solutions, Inc. (2013-12-04). "LCD L32752 – Transcranial Magnetic Stimulation for Depression". Contractor's Determination Number L32752. Centers for Medicare and Medicaid Services. Retrieved 2014-02-17.
- ^ Novitas Solutions, Inc. (2013-12-05). "LCD L33660 – Transcranial Magnetic Stimulation (TMS) for the Treatment of Depression". Contractor's Determination Number L33660. Centers for Medicare and Medicaid Services. Retrieved 2014-02-17.
- ^ Benussi A, Pascual-Leone A, Borroni B (March 2020). "Non-Invasive Cerebellar Stimulation in Neurodegenerative Ataxia: A Literature Review". Int J Mol Sci. 21 (6): 1948. doi:10.3390/ijms21061948. PMC 7139863. PMID 32178459.
- ^ Rosa MA, Lisanby SH (January 2012). "Somatic treatments for mood disorders". Neuropsychopharmacology. 37 (1): 102–16. doi:10.1038/npp.2011.225. PMC 3238088. PMID 21976043.
- ^ Grimaldi G, Argyropoulos GP, Boehringer A, Celnik P, Edwards MJ, Ferrucci R, Galea JM, Groiss SJ, Hiraoka K, Kassavetis P, Lesage E, Manto M, Miall RC, Priori A, Sadnicka A, Ugawa Y, Ziemann U (February 2014). "Non-invasive cerebellar stimulation—a consensus paper". Cerebellum. 13 (1): 121–38. doi:10.1007/s12311-013-0514-7. PMID 23943521.
- ^ Siebner HR, Hartwigsen G, Kassuba T, Rothwell JC (October 2009). "How does transcranial magnetic stimulation modify neuronal activity in the brain? Implications for studies of cognition". Cortex. 45 (9): 1035–42. doi:10.1016/j.cortex.2009.02.007. PMC 2997692. PMID 19371866.
- ^ a b Sparing R, Mottaghy FM (April 2008). "Noninvasive brain stimulation with transcranial magnetic or direct current stimulation (TMS/tDCS)-From insights into human memory to therapy of its dysfunction". Methods. 44 (4): 329–37. doi:10.1016/j.ymeth.2007.02.001. PMID 18374276.
